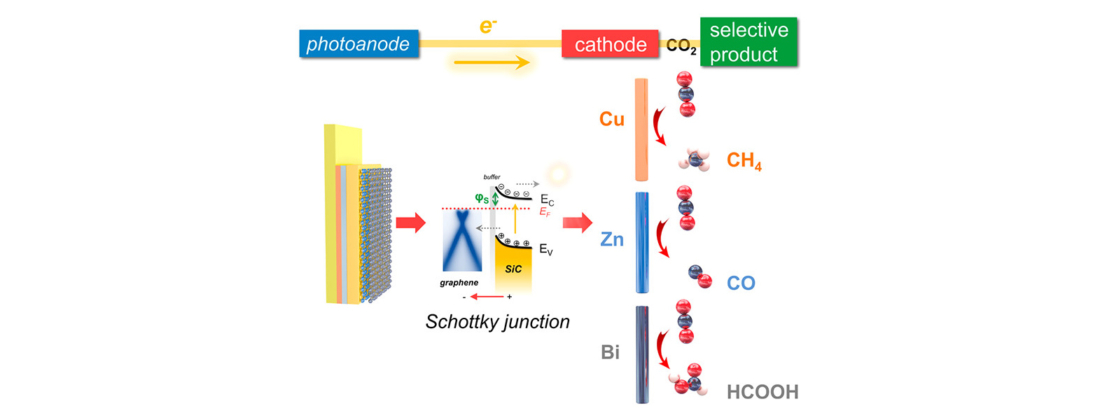
Researchers at Linköping University in Sweden are developing a promising new method to selectively convert carbon dioxide and water to various types of fuel. Driving this reaction is solar energy. The recent study, published in ACS Nano, combines the material graphene and the semiconductor cubic silicon carbide in a process which essentially mimics photosynthesis in plants.
“Our work has demonstrated that the graphene-cubic silicon carbide material structure shows a great potential to realize solar-driven conversion of CO2 and water into chemical fuels. Our long-term objective is to enhance the solar energy conversion efficiency in order to commercialize this technology,” said Jianwu Sun, senior lecturer at Linköping University and principal investigator of the study.
Deep, elemental understanding of photosynthesis has been a long-sought goal of the science community, and a new exploration for technology innovators. Cross-discipline work can result in sustainable solutions or applications which incorporate characteristics of biomimicry, a design re-creation of biological processes and materials in nature. In structural studies, graphene, one of the thinnest materials in the known universe, is valued for its versatility for application in biomedical, optical, and photoelectrochemical devices.
Imaging graphene with MAXPEEM
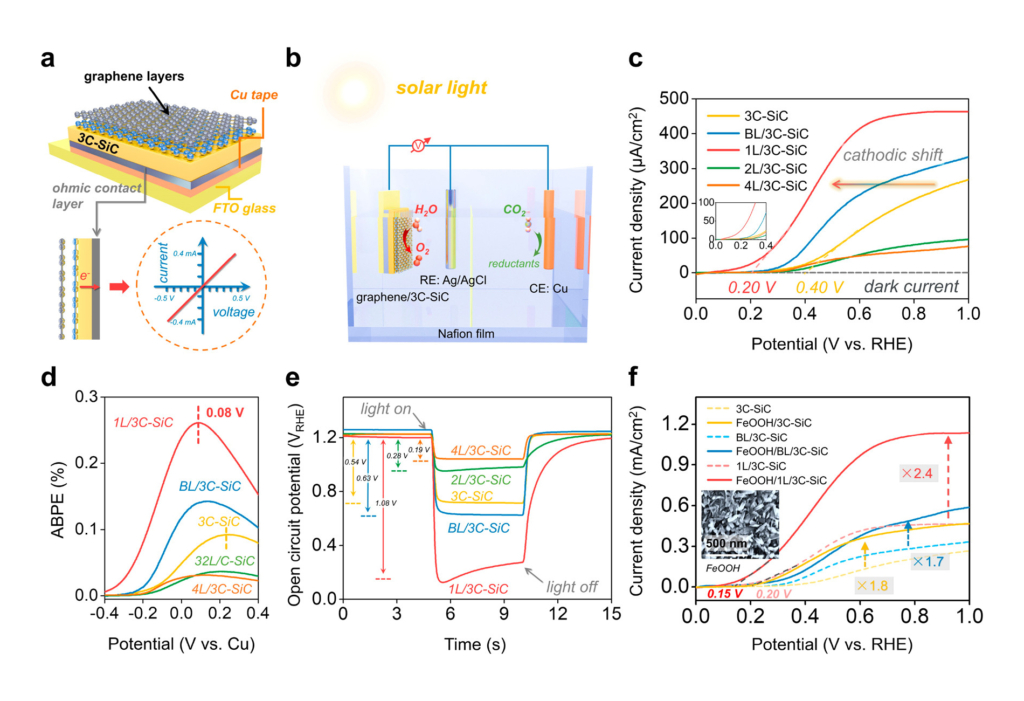
The study investigated how to engineer a tunable graphene-semiconductor interface while conserving graphene’s desired properties of stability, flexibility, elasticity, and conductivity. Sun’s research group constructed a viable photoelectrode with adjustable Schottky junction. The photoelectrode was developed by growing a tailored number of uniform graphene layers on cubic silicon carbide (3C-SiC) semiconductor, the latter of which captures sunlight and forms an inherent electric field with graphene. Combined with select metal cathodes, this field allows for charge carriers to activate a photoelectrochemical reaction—that is, to generate fuel—with the application of solar energy.
The reaction was tested using a two-compartment photoelectrochemical (PEC) cell separated by a Nafion membrane for water oxidation and CO2 reduction. The study selectively produced fuel sources such as methane, pure carbon monoxide and formic acid.
The Jianwu Sun Research Laboratory is part of IFM, the Department of Physics, Chemistry and Biology at Linköping University. Low-energy electron microscopy (LEEM) and low-energy electron diffraction (LEED) characterizations on graphene were performed at the surface imaging beamline MAXPEEM at MAX IV.
In the development of this technique, the team acquired some unexpected insights into the material function. “We found that the graphene/3C-SiC material structure shows a synergistic enhancement of the device stability and efficiency. Graphene is highly transparent and conductive. These properties allow for fully sunlight transmission and enable a fast charge transfer for photoelectrochemical CO2 conversion. Meanwhile, graphene also protects 3C-SiC surface against corrosion, thus enhancing the stability,” explained Sun.
This work advances previous studies by Sun and colleagues which focus on different aspects of graphene and SiC for conversion of solar energy into chemical fuels. For example, the lab developed a key method to grow graphene with different thicknesses on cubic silicon carbide in a controlled manner.
Sun’s lab is currently focused on further studies of semiconductors graphene and SiC for conversion of solar energy into fuels. The research involves use of 3C-SiC and graphene for hydrogen (H2) generation via solar-driven water splitting. The project is supported by the Swedish Research Council, FORMAS, and the Swedish Foundation for International Cooperation in Research and Higher Education.